Measure microscopes performance & detect issues
You don’t know if you don’t measure it.
Troubleshooting microscopes or performing quality control starts with you having the relevant information. You need reliable information, quickly! so you can take the right action and save the situation.
But time-efficiency does not mean cutting back on information. We give you the best information/time ratio possible from a quick 5 minutes check or a 1 hour in-depth performance assessment.
Discover Argolight Troubleshooting and Quality Control software, DAYBOOK
Daybook works with Argolight hardware solutions
Analyze
More than 12 automated quality analyses, several tens of relevant metrics.
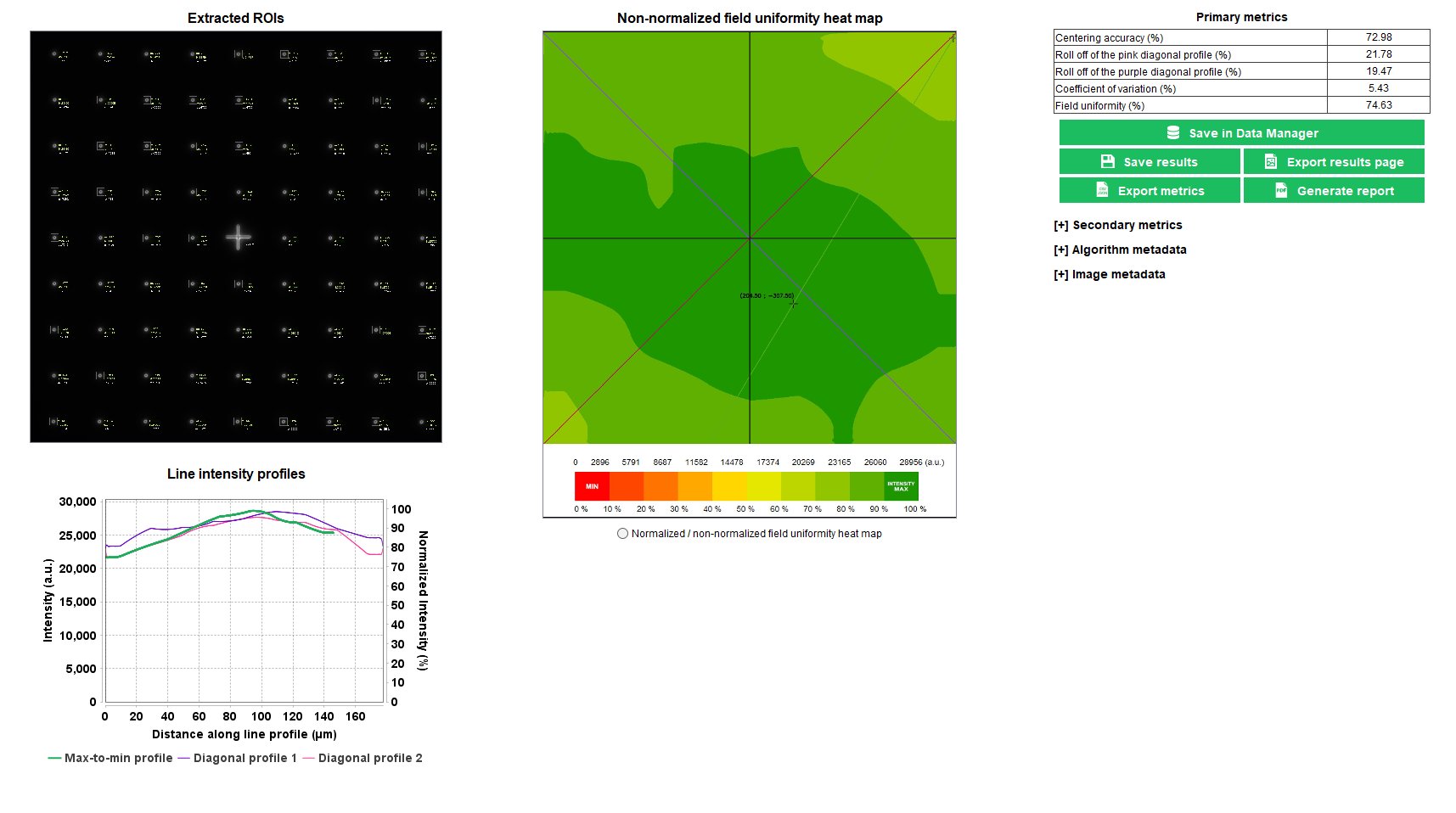
Field uniformity
The field uniformity is a well-known parameter related to the inhomogeneous spatial distribution of the intensity in the image of an even object.
In any fluorescence microscope, the knowledge of the field uniformity is important when the image intensity information is aimed to be measured. For intensity quantification in images of biological samples, the field uniformity shall be known, and eventually corrected, to have access to accurate measurements.
The “field uniformity” analysis provides the spatial distribution of the intensity, due to illumination and detection inhomogeneities, within the field of view.
Matrix of Rings pattern available on

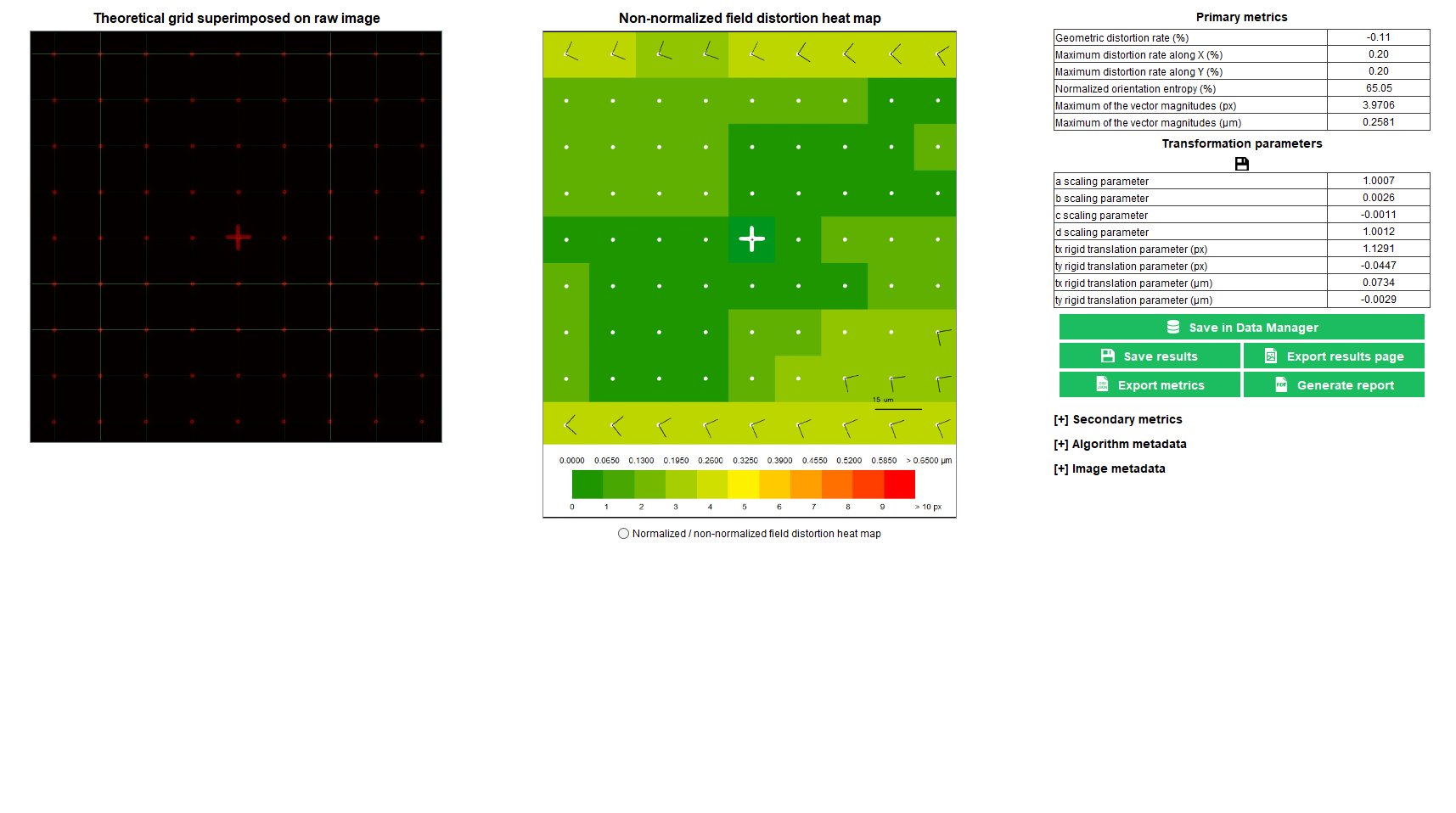
Field Distortion
The field distortion is an optical aberration inducing a spatial deformation of the imaged object, usually at the corners of the image. In the presence of field distortion, the magnification is not constant over the field of view; it is dependent on the XY coordinates.
In any fluorescence microscope, the knowledge of the field distortion is important when spatial information in an image is aimed to be measured. For positions, lengths, areas or volumes quantification in images of biological samples, the field distortion shall be known, and eventually corrected, to have access to accurate measurements.
The “field distortion” analysis provides the lateral shifts, due to distortion, within the field of view.
Matrix of Rings pattern available on

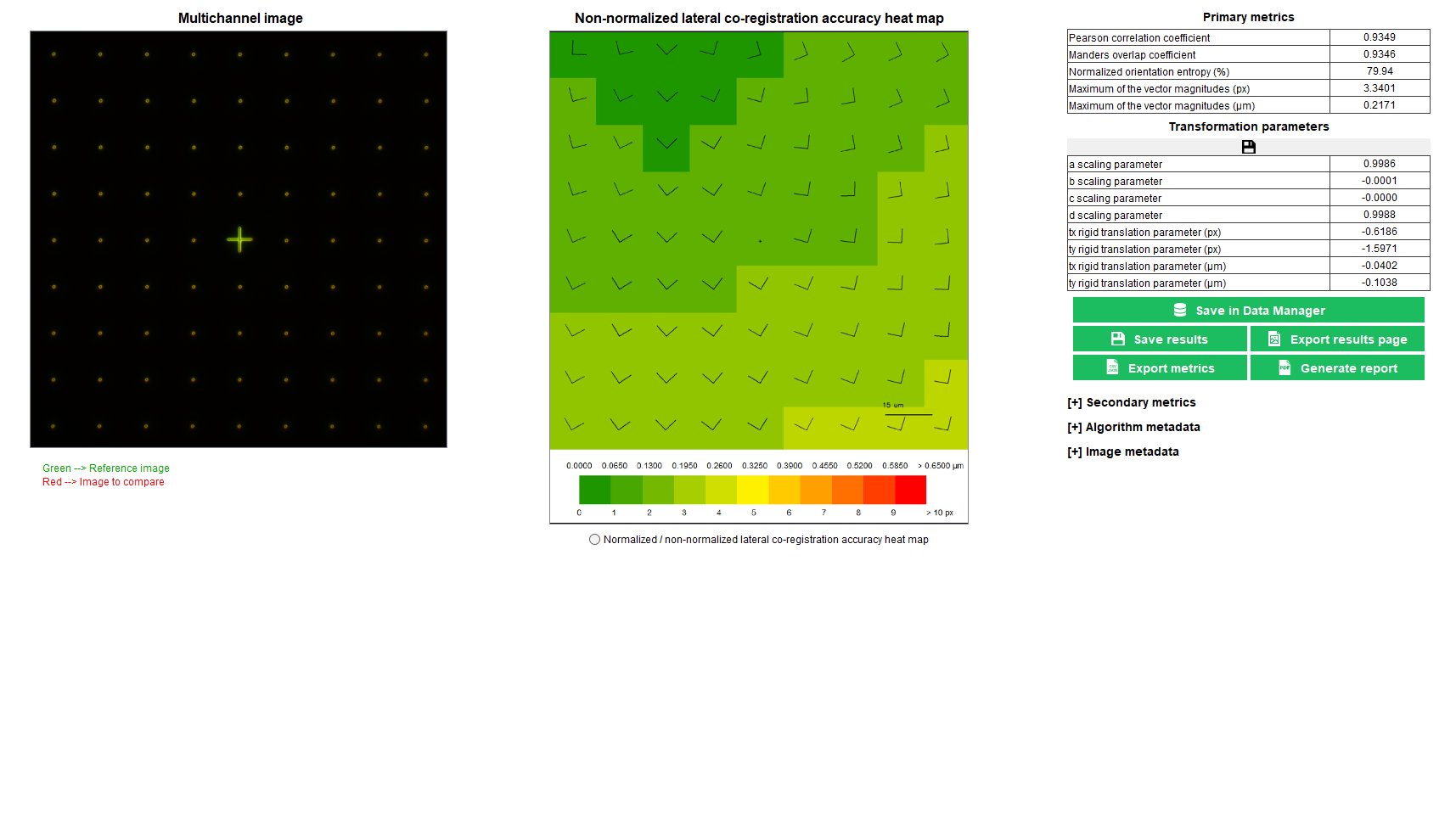
Lateral co-registration accuracy
The co-registration accuracy is the ability of a fluorescence microscope to record images of a multi-labeled object without introducing additional shifts that would not be related to the object itself.
In any fluorescence microscope, the knowledge of the co-registration accuracy, both in the lateral and axial directions, is important when color information in an image is aimed to be used. For co-localization quantification in images of biological samples stained with several labels, the co-registration accuracy between the different channels shall be known, and eventually corrected, to prevent from misinterpretation.
The “lateral co-registration accuracy” analysis provides the lateral shifts between two channels, due to the co-registration inaccuracy of the system, within the entire field of view.
Matrix of Rings pattern available on

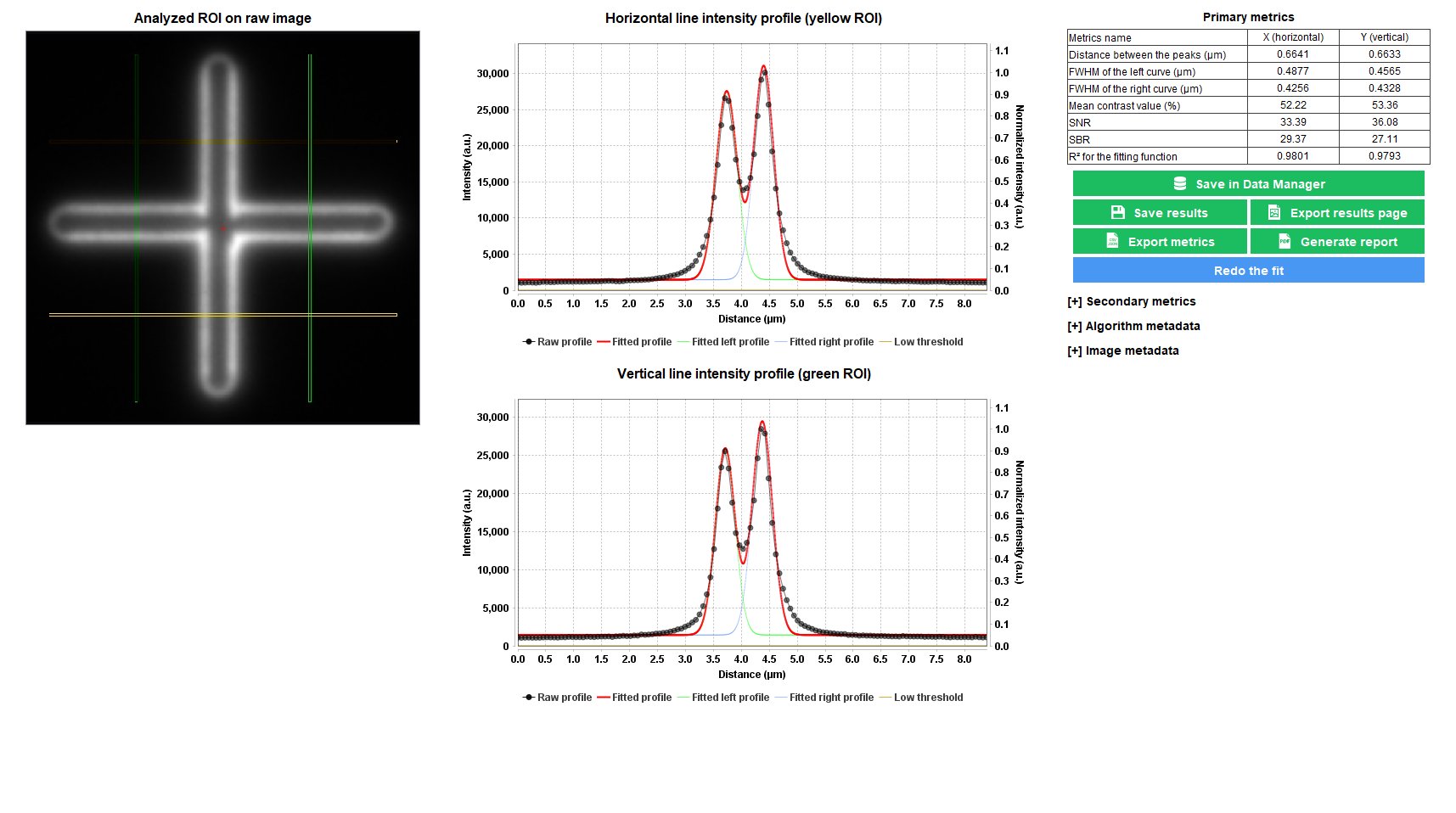
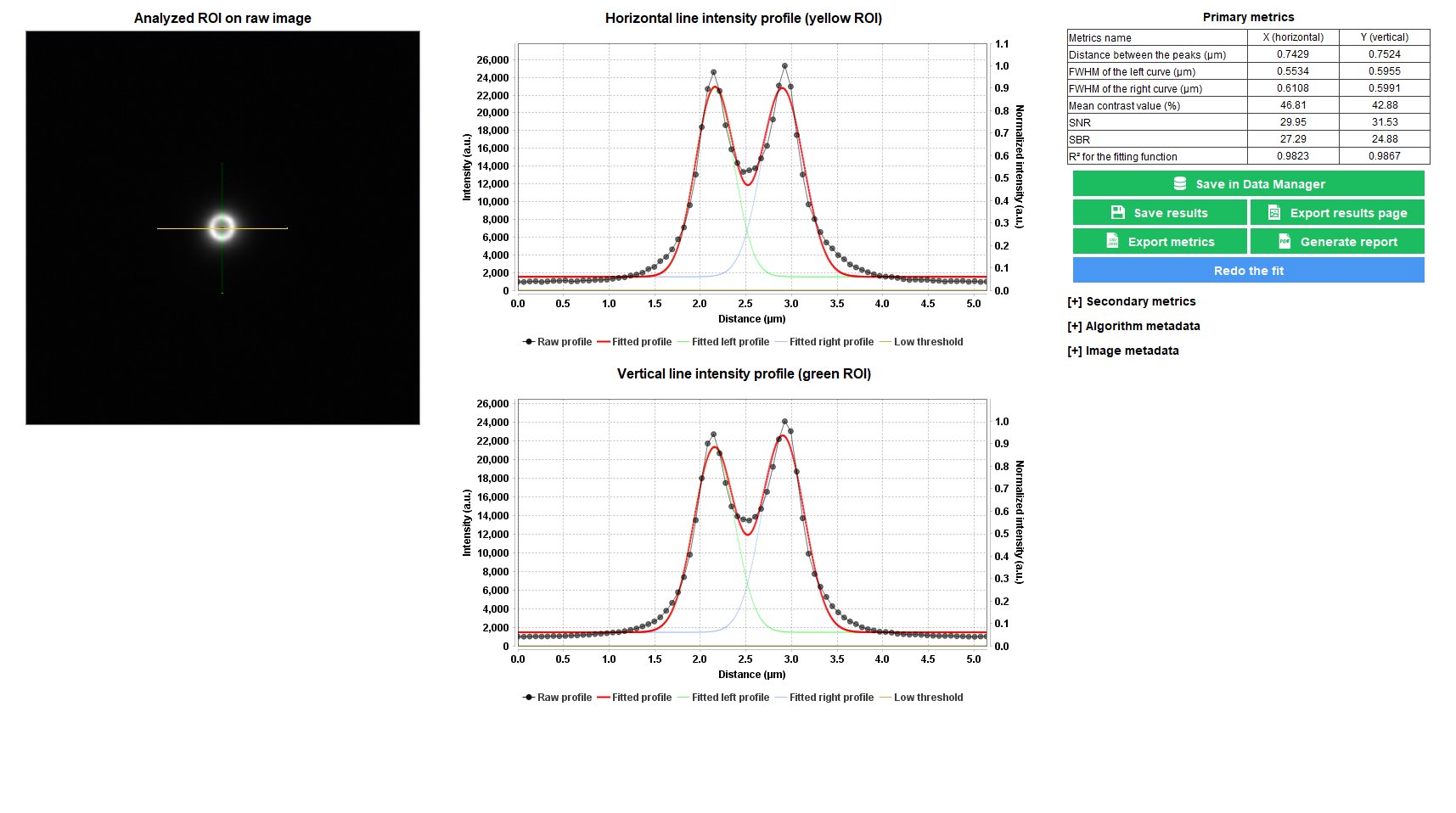
Line Spread Function
The line spread function (LSF) gives access to the spreading behavior of the light by a line object. Fitting the LSF with a mathematical function (double Gaussian, double Lorentzian, double Sech²) allows to extract the full width at half maximum (FWHM), which is a parameter related to the lateral resolution of the imaging system.
The “line spread function” analysis provides information, in the XY plane, on how light spreads from a line, as well as quantitative parameters such as the FHWM of the LSF and the signal-to-noise ratio (SNR) value in the image.
Matrix of Rings pattern available on

Ring Spread Function
The ring spread function (RSF) gives access to the spreading behavior of the light by a ring object. Fitting the RSF with a mathematical function (double Gaussian, double Lorentzian, double Sech²) allows to extract the full width at half maximum (FWHM), which is a parameter related to the lateral resolution of the imaging system.
The “ring spread function” analysis provides information, in the XY plane, on how light spreads from a ring, as well as quantitative parameters such as the FHWM of the RSF and the signal-to-noise ratio (SNR) value in the image.
Matrix of Rings pattern available on

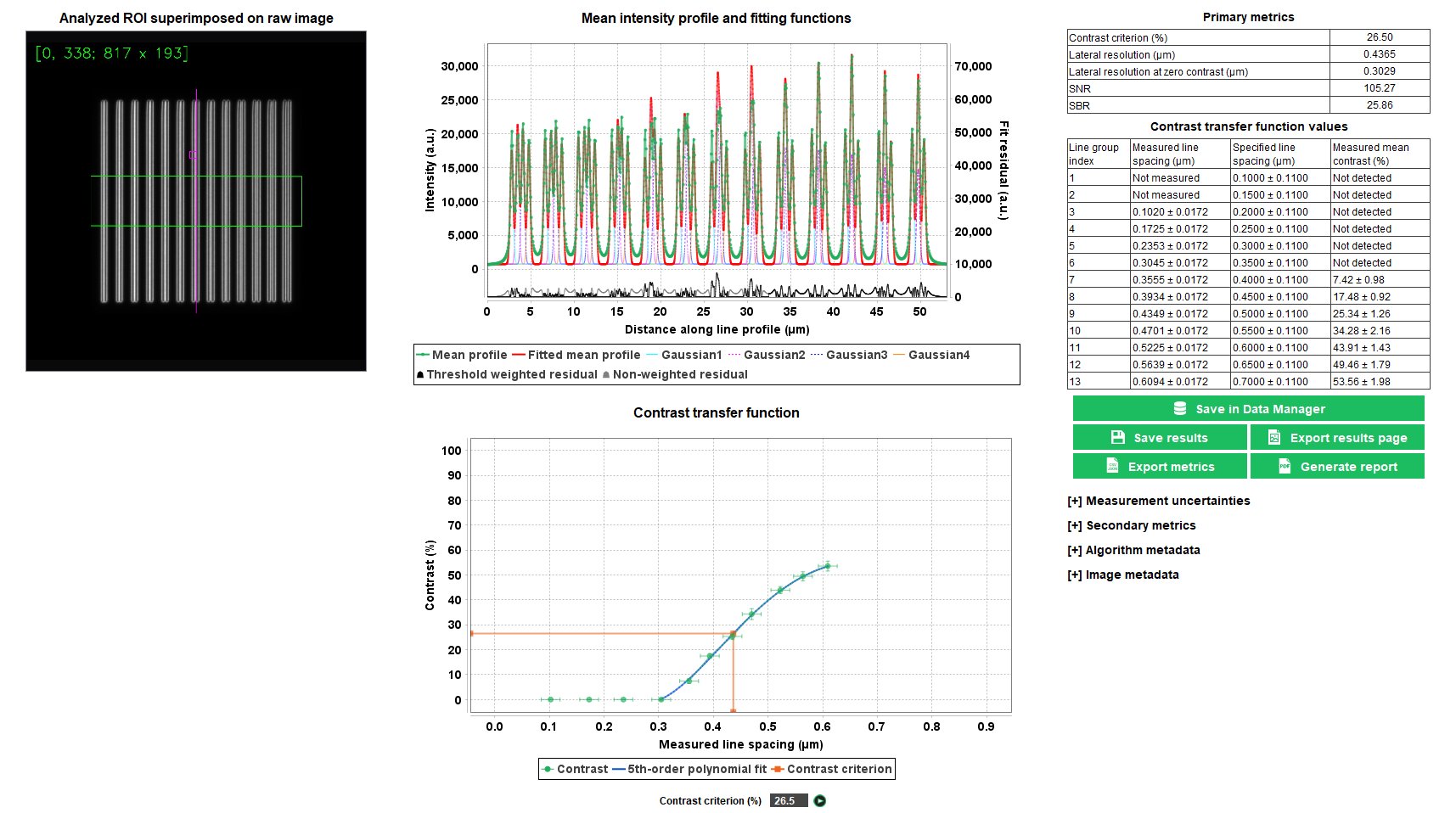
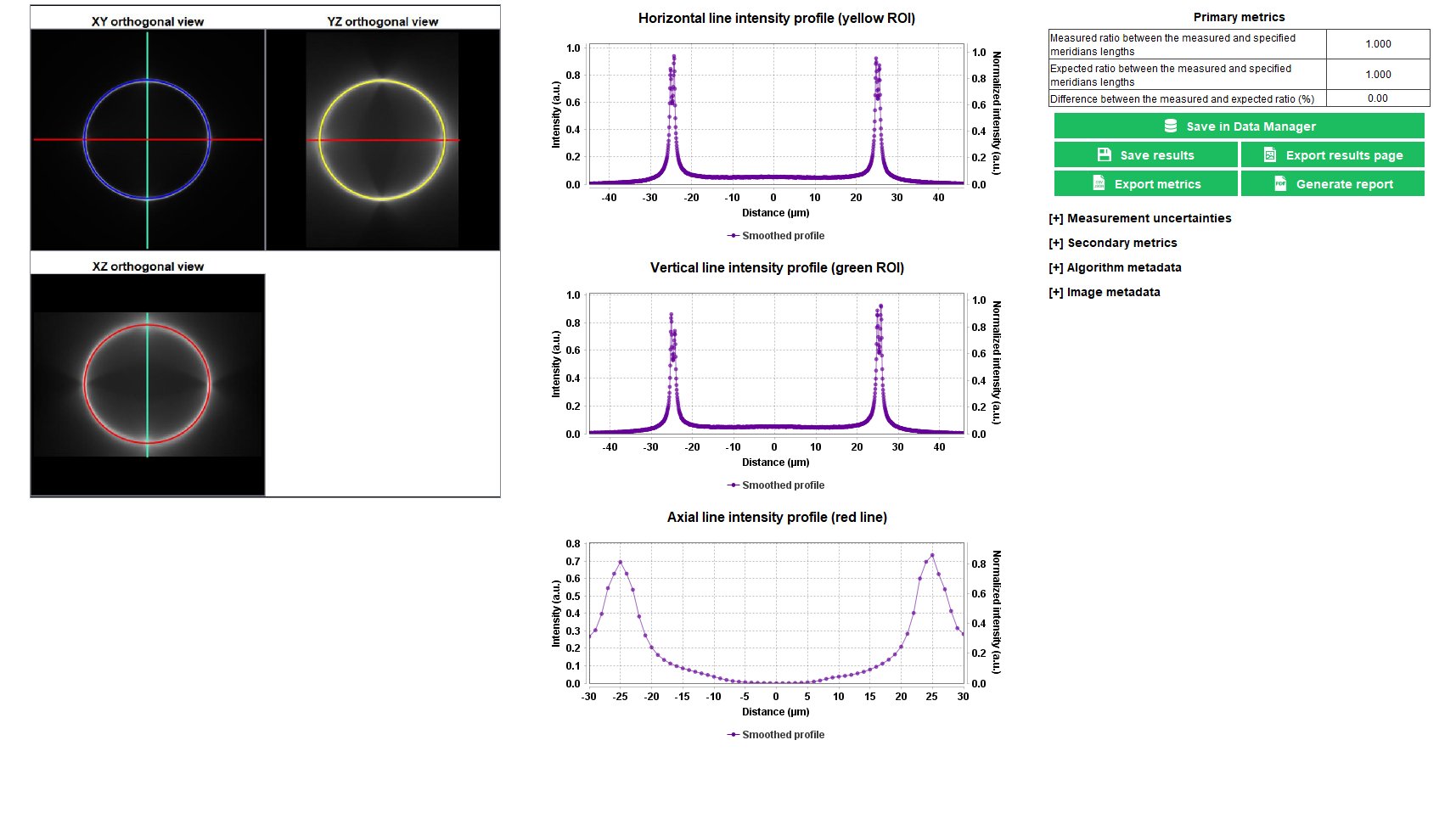
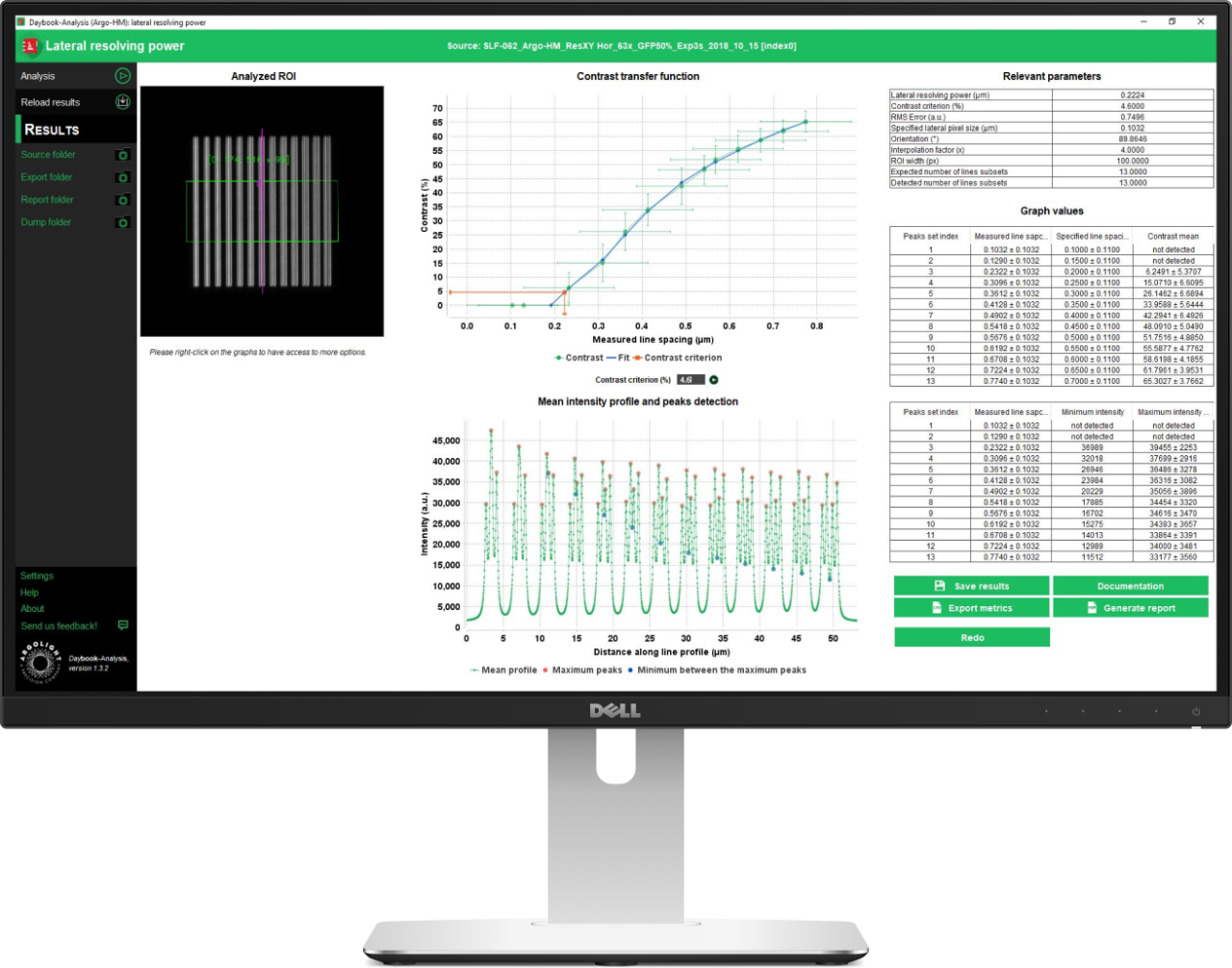
Lateral Resolution
The lateral resolution is the minimal distance between two objects close to each other that an imaging system can clearly measure. There are different ways to measure the lateral resolution. The method used in this analysis relies on the contrast transfer function, i.e. the measured contrast vs the measured distance between gradually spaced lines.
The “lateral resolution” analysis provides the minimal resolvable distance between lines close to each other, for a given contrast value, with an associated signal-to-noise ratio (SNR) value in the image.
Gradually Spaced Lines pattern available on

Stage Repositioning Repeatability
During the acquisition of image tiles, the lateral stage may not reposition to some location in a repeatable way. Therefore, it is important to ensure that the XY stage repositioning repeatability is sufficiently accurate when tiling and stitching large regions in 2D or 3D.
The “stage repositioning repeatability” analysis allows to determine the XY motorized stage repositioning repeatability after many round trips, to ensure it is within the specifications.
Repositioning Crosses pattern available on
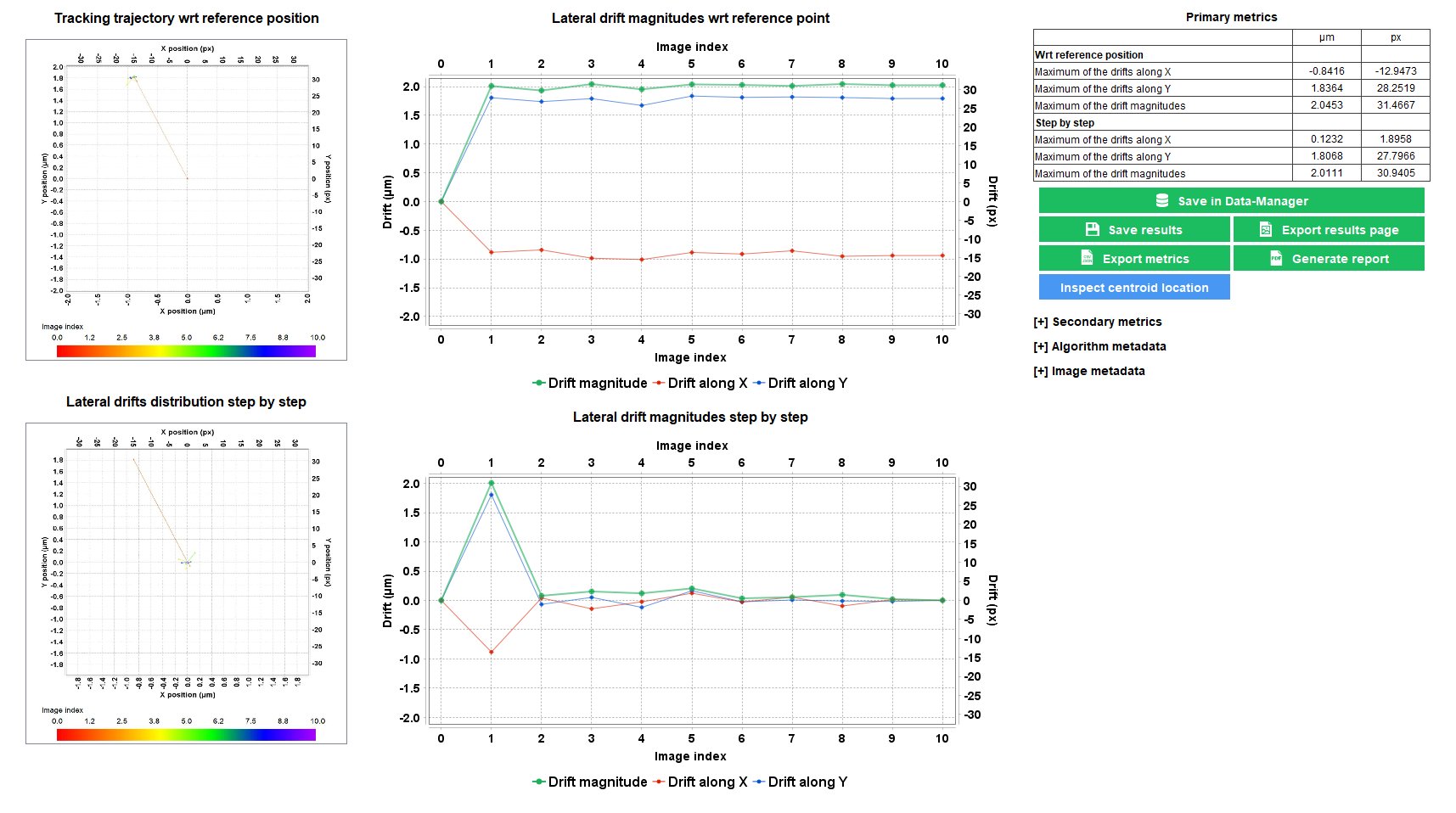
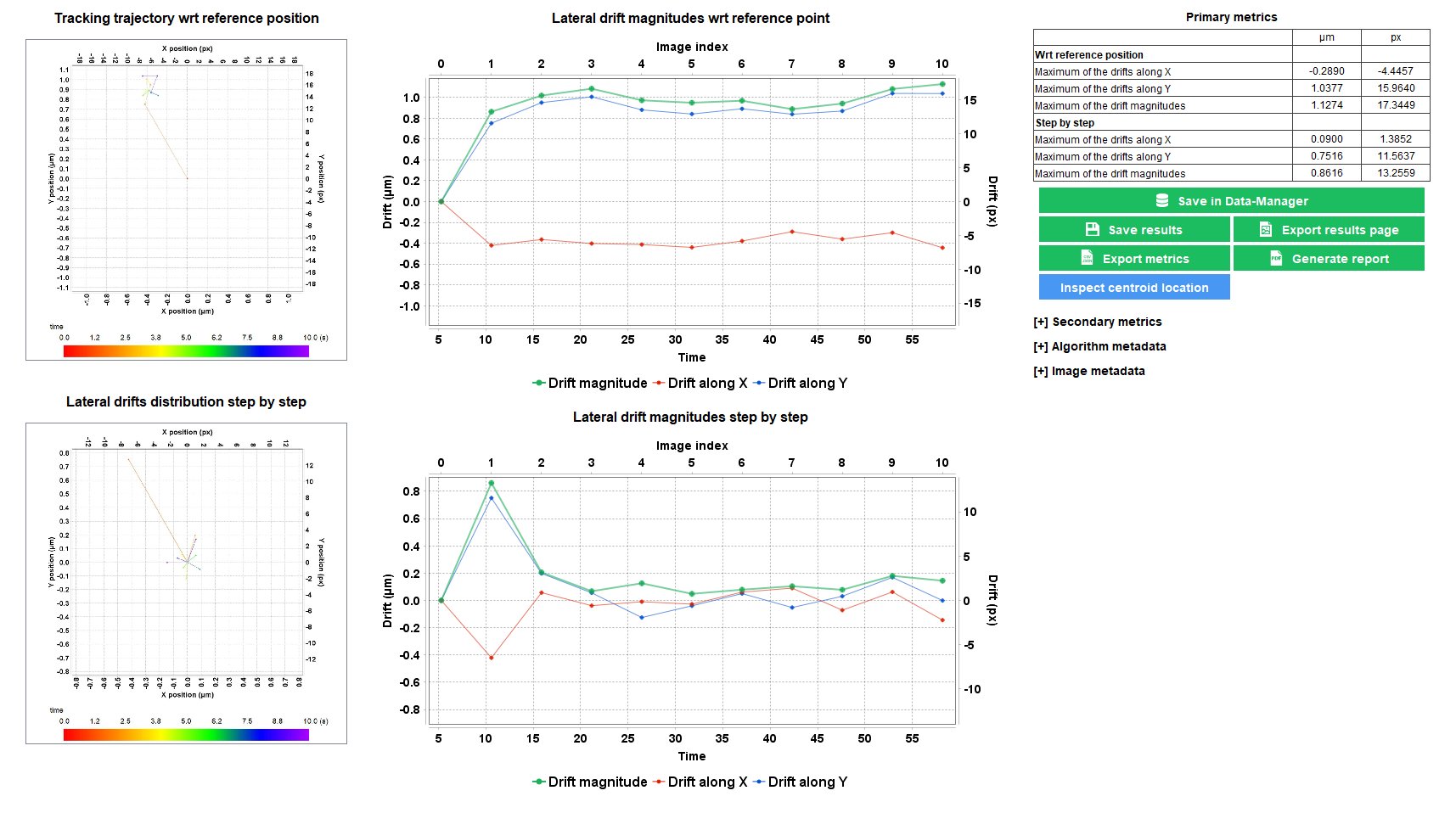
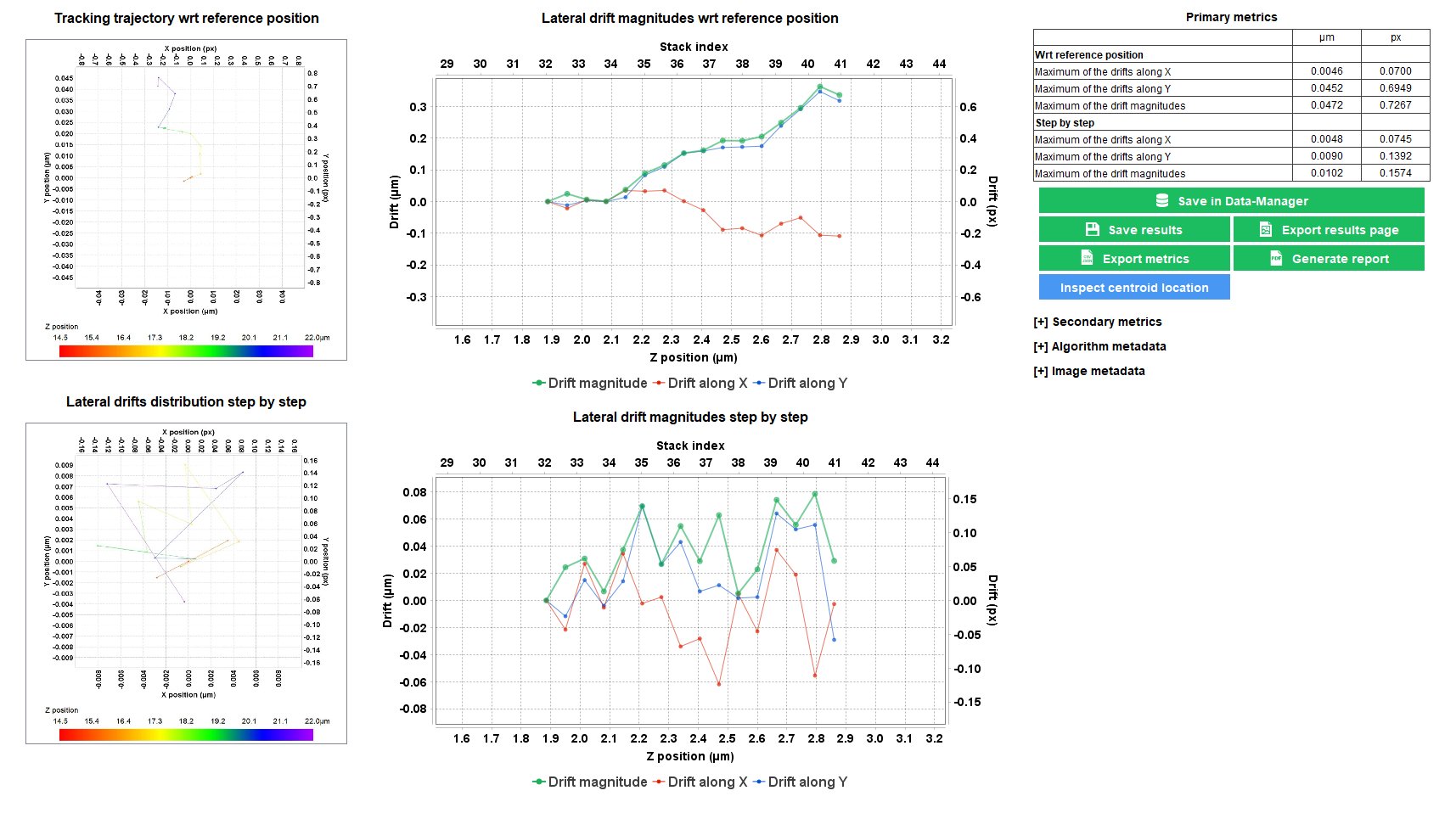
Stage Drift During Timelapse
When performing timelapse imaging, it is important to ensure that the XY stage do not drift too much. Sources of stage drift can origin from the environmental conditions, such as temperature fluctuations and air flow.
The “stage drift during timelapse” analysis allows to measure the XY stage drift during a timelapse imaging, to ensure it is within the specifications.
Repositioning Crosses pattern available on
Stage Drift During Z Stacking
This test concerns only the microscopes equipped with a motorized Z stage.
During the acquisition of image Z-stacks, the lateral (XY) stage may drift. Therefore, it is important to ensure that the XY stage do not drift too much. Sources of stage drift can origin from the environmental conditions, such as temperature fluctuations and air flow.
The “stage drift during Z-stacking” analysis allows to determine the XY stage drift during Z-stacking, to ensure it is within the specifications.
Accuracy of 3D reconstruction
This test concerns only the microscopes equipped with a motorized Z stage.
Confocal fluorescence microscopes allow to perform 3D imaging, thanks to their ability to remove the out-of-focus light. 3D imaging is achieved by reconstructing a Z-stack of images, which accuracy may be altered by different issues. Sources of 3D reconstruction inaccuracy can origin from the Z-stage, from a wrong adjustment of the objective correction collar for coverslip thickness or from the environmental conditions, such as temperature fluctuations and air flow.
The “accuracy of 3D reconstruction” analysis allows to check the ability of confocal fluorescence microscopes to acquire Z-stacks of images correctly, so that their reconstruction into 3D images is accurate, i.e. representative of the reality.
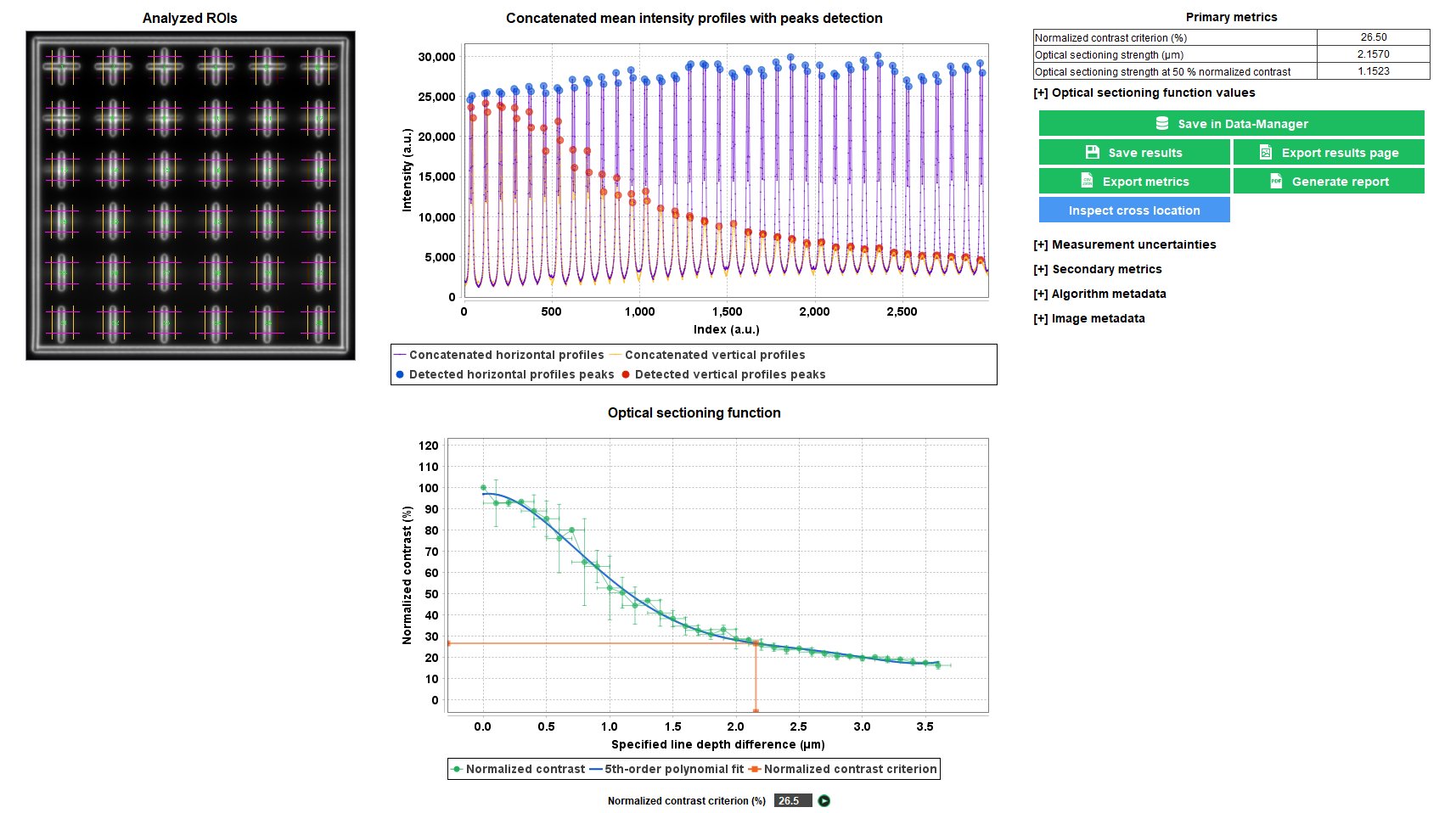
Optical sectioning strength
The optical sectioning is the ability of an imaging system to acquire clear slices of a thick object, in order to reconstruct it in 3D from images captured at different focal plane. There are different ways to measure the optical sectioning strength. The method used in this analysis relies on the optical sectioning function, i.e. the measured contrast versus the specified distance between lines on the same plane and lines going deeper and deeper in the glass.
The “optical sectioning” analysis provides the optical sectioning strength between lines at different depths, for a given contrast value.
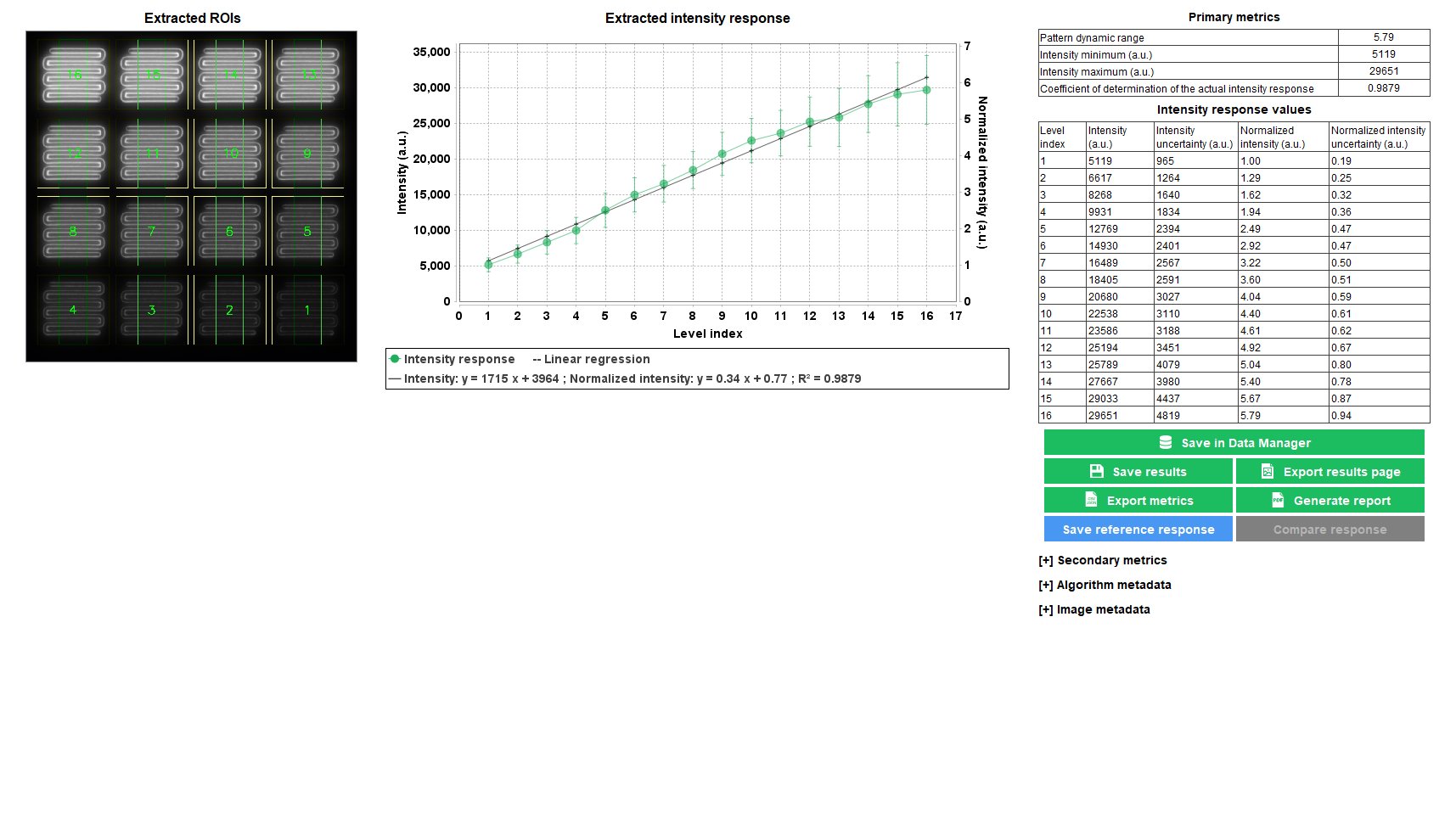
Intensity Response
The intensity response of a fluorescence microscope expresses the output digital signal to an input photon flux. It depends on many aspects of the microscope, either in the illumination path or the detection path.
In any fluorescence microscope, the overall intensity response may evolve over time. For example, variations in the illumination power, detector sensitivity, optical alignment, etc. can lead to intensity response fluctuations. The knowledge of the intensity response is therefore important when the intensity quantification in images of a biological sample, acquired at different times, is aimed to be performed.
The “intensity response” analysis provides the intensity response of the imaging system to 16 intensity levels following a linear evolution, as well as quantitative parameters such as intensity values and pattern dynamic range. Monitoring these parameters allows to observe how the intensity response evolves over time, with respect to reference values.
4x4 or 2x16 Intensity pattern available on
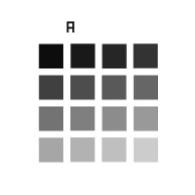
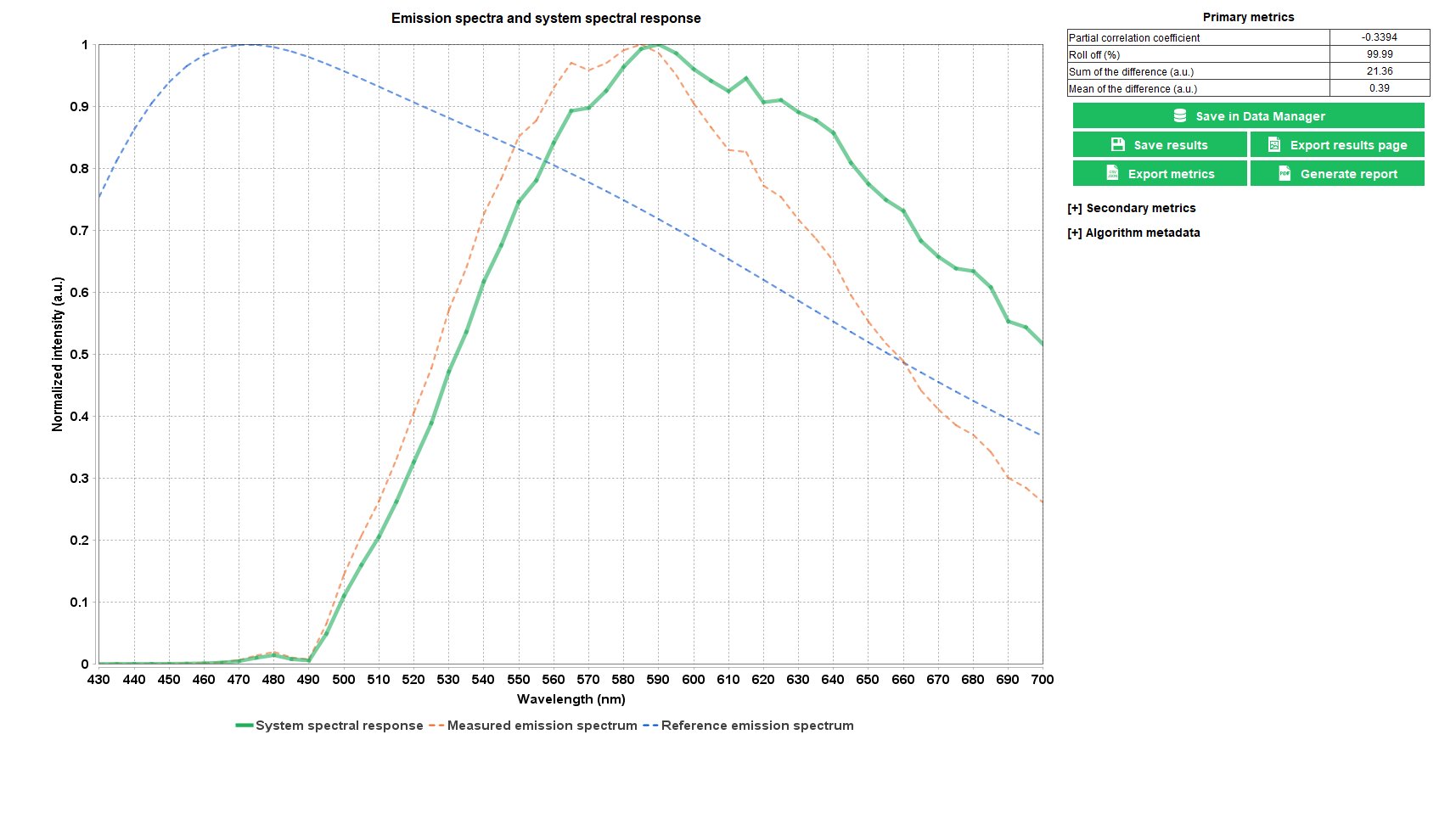
Spectral Response
This test concerns only the microscopes that can acquire fluorescence emission spectra.
Fluorescence microscopes, although conceived preliminary to be imaging systems, can also acquire emission spectra, with a better spatial resolution but poorer spectral resolution than conventional spectrometers. The spectral response of a microscope expresses the responsivity of the imaging system versus the emission wavelengths.
Correct interpretation of the emission spectra measured with a microscope requires the knowledge of its spectral response.
The “spectral response” analysis provides the normalized detection efficiency versus the emission wavelengths, as well as quantitative parameters such as the spectral roll-off.
Argolight logo pattern available on

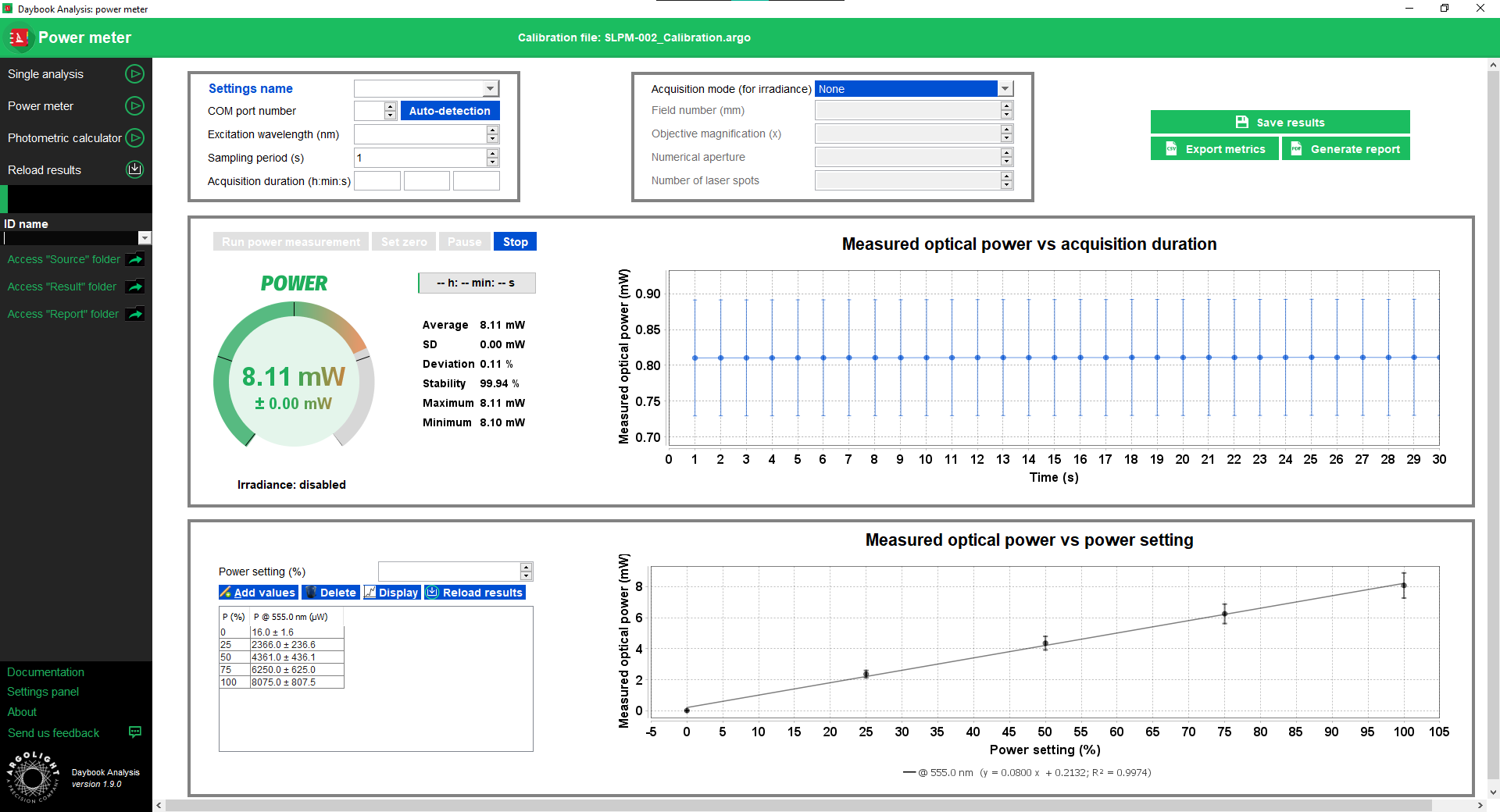
Power Meter
This test concerns only the Argo-POWER slide.
The optical power measured at the sample plane is a key parameter in microscopy setups, either to troubleshoot any issue or to perform quantitative microscopy.
Measuring the optical power at regular interval using the same illumination settings allows to troubleshoot, for example, changes in light sources power, drops in filters transmission, or laser-to-fiber coupling misalignment.
Measuring the optical power prior and after an imaging campaign ensures that the light source power has not fluctuated in the meantime, evidencing it has not affected the intensity values in the images.
Measuring the optical power prior an imaging campaign using the same illumination settings as for a previous imaging campaign ensures that the same optical power is applied on the sample, so that the illumination conditions are reproduced.
Measuring the optical power allows to report this value in scientific publications, so that other researchers can reproduce these illumination conditions, thus contributing to the combat against the replication crisis.
The “power meter” analysis allows to perform live optical power, live irradiance, time-lapse and power vs setting measurements.
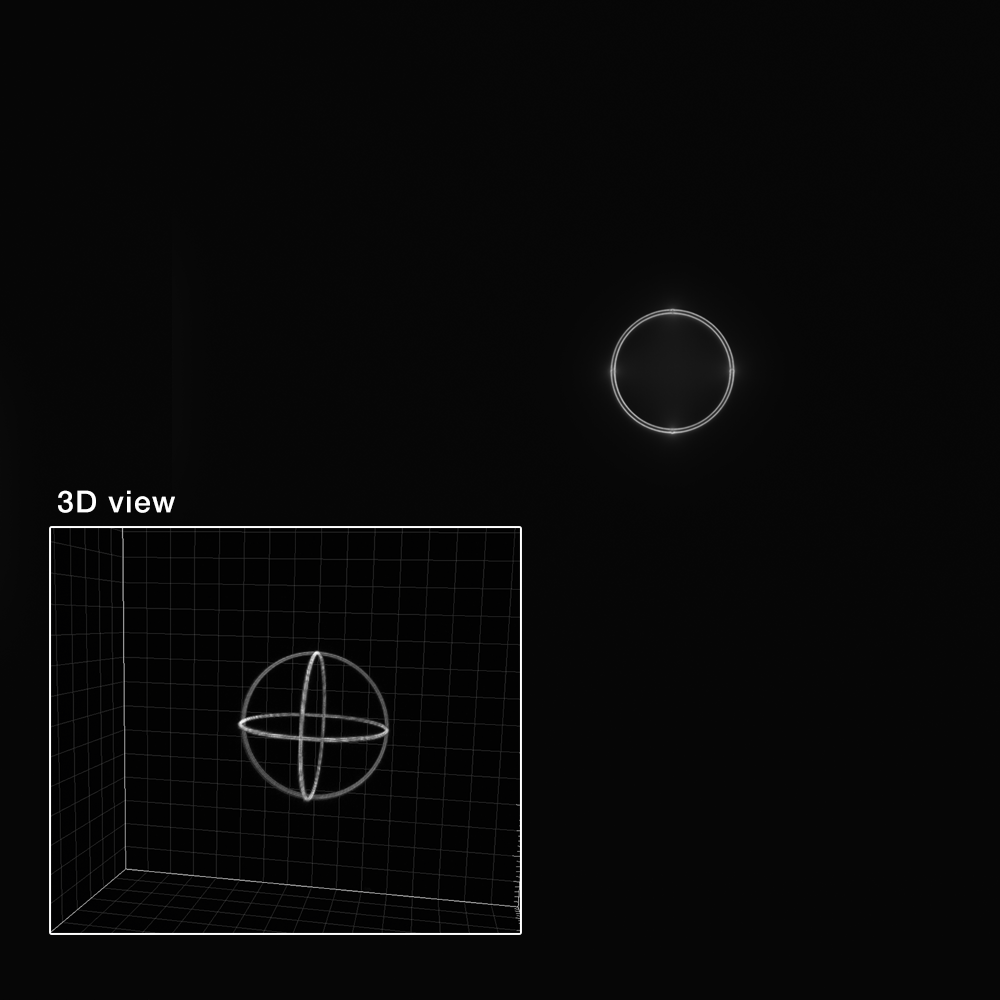
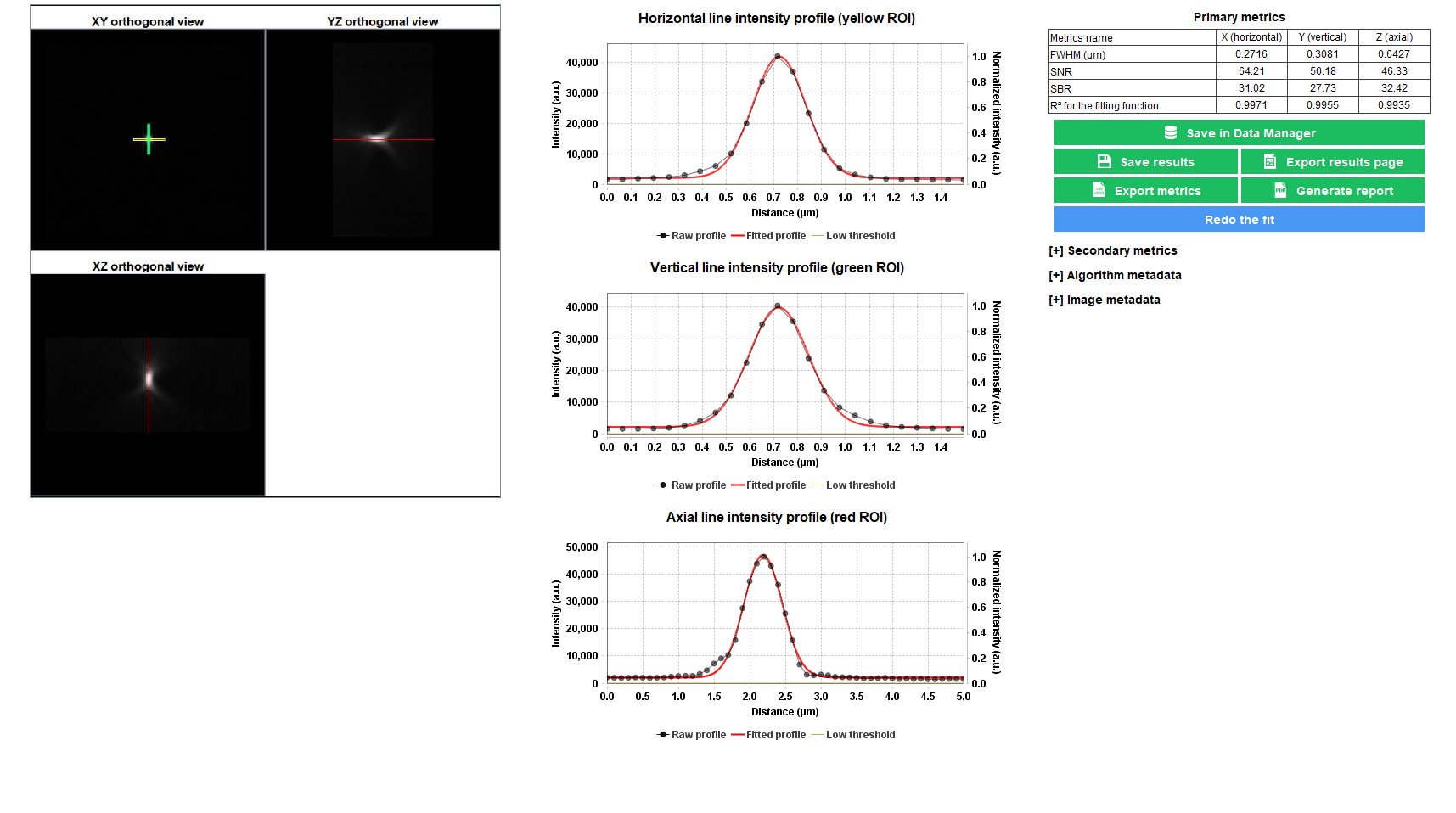
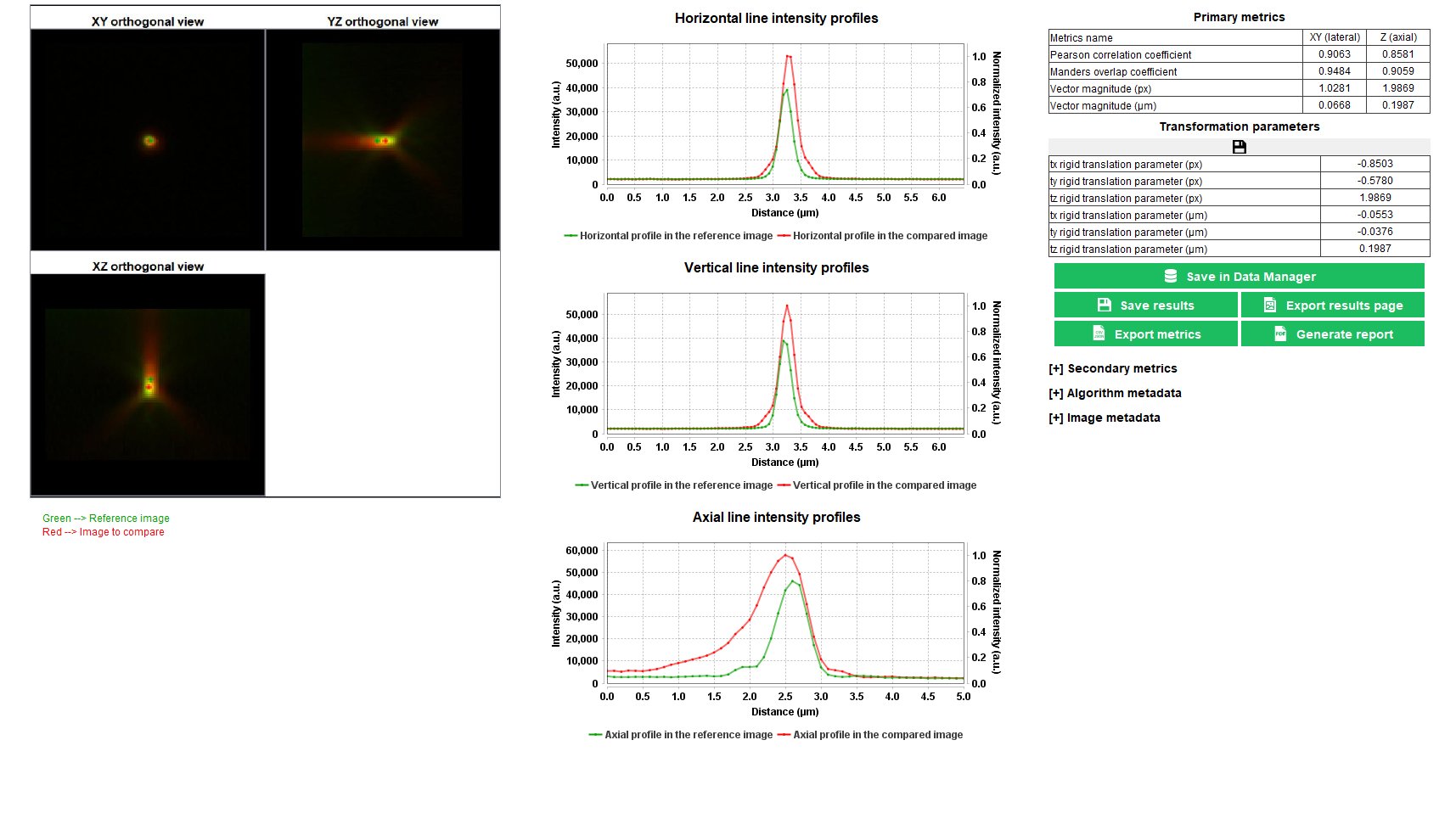
Point Spread Function
The point spread function (PSF) gives access to the spreading behavior of the light by a point-like object. Fitting the PSF with a mathematical function (Gaussian, Lorentzian, Sech²) allows to extract the full width at half maximum (FWHM) along X, Y and Z, which is a parameter that commonly describes the resolution of the imaging system.
The “point spread function” analysis provides information, in 3D, on how light spreads from a point-like object, as well as quantitative parameters such as the lateral and axial FHWM of the PSF and the signal-to-noise ratio (SNR) in the image.
Accuracy of co-registration
This test concerns only the microscopes equipped with a motorized Z stage.
The co-registration accuracy is the ability of a fluorescence microscope to record images of a multi-labeled object without introducing additional shifts that would not be related to the object itself.
In any fluorescence microscope, the knowledge of the co-registration accuracy, both in the lateral and axial directions, is important when color information in an image is aimed to be used. For co-localization quantification in images of biological samples stained with several labels, the co-registration accuracy between the different channels shall be known, and eventually corrected, to prevent from misinterpretation.
The “accuracy of co-registration” analysis provides the lateral and axial shifts between two channels, that are due to the translation-only contribution to the co-registration inaccuracy of the system, within a limited area of the field of view.
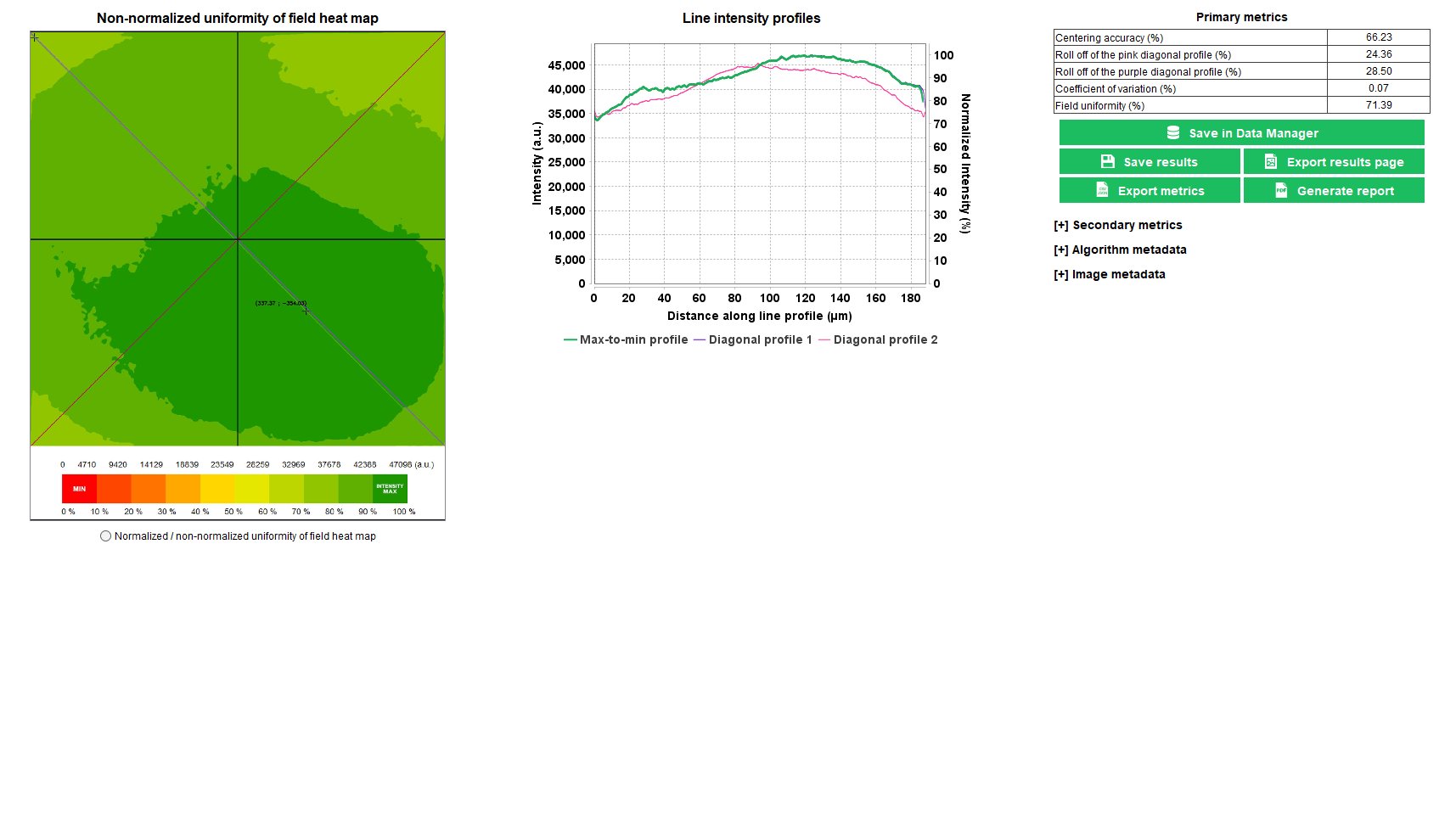
Uniformity of field
The uniformity of field is a well-known parameter related to the inhomogeneous spatial distribution of the intensity in the image of an even object.
In any fluorescence microscope, the knowledge of the uniformity of field is important when the image intensity information is aimed to be measured. For intensity quantification in images of biological samples, the uniformity of field shall be known, and eventually corrected, to have access to accurate measurements.
The “uniformity of field” analysis provides the spatial distribution of the intensity, due to illumination and detection inhomogeneities, within the field of view.
Detect
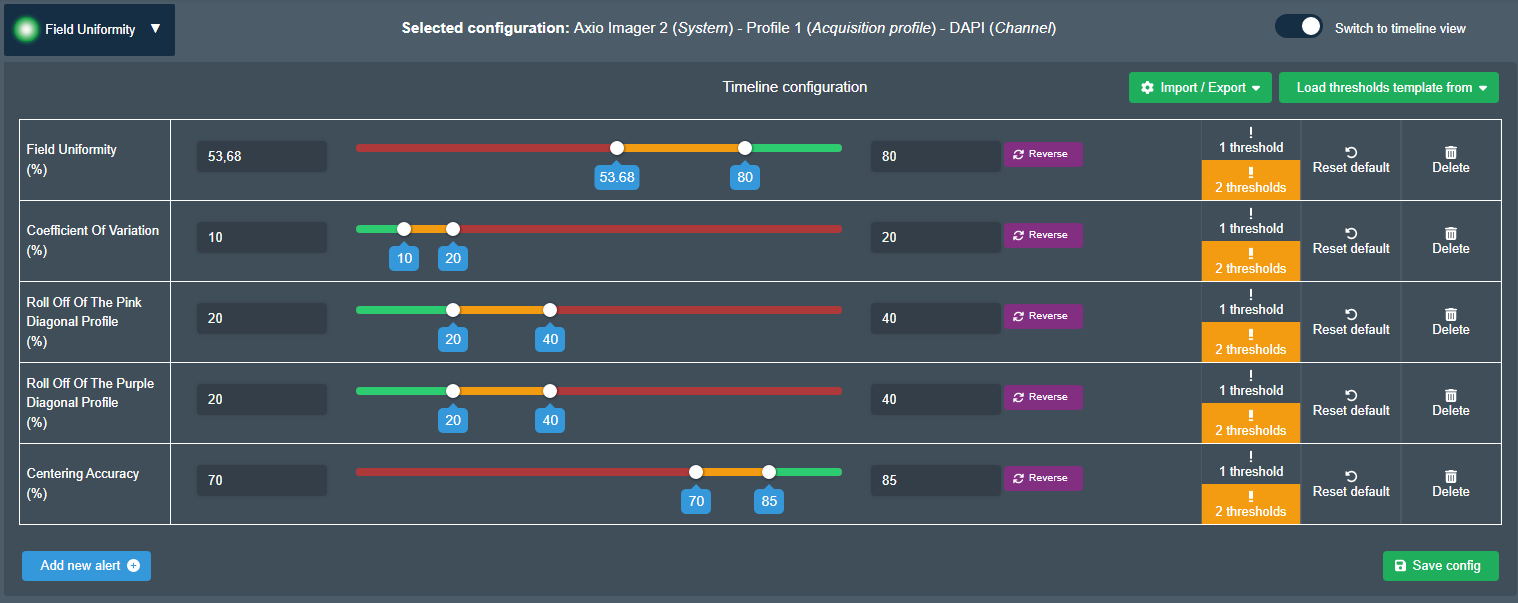
Set tolerance thresholds
You are not efficient when you spend time demonstrating that systems work well.
Quality thresholds are the solution. Show right away that systems are performing and focus on the real issues.
You can select one or two threshold values and the orientation of the traffic light colors.
Automate
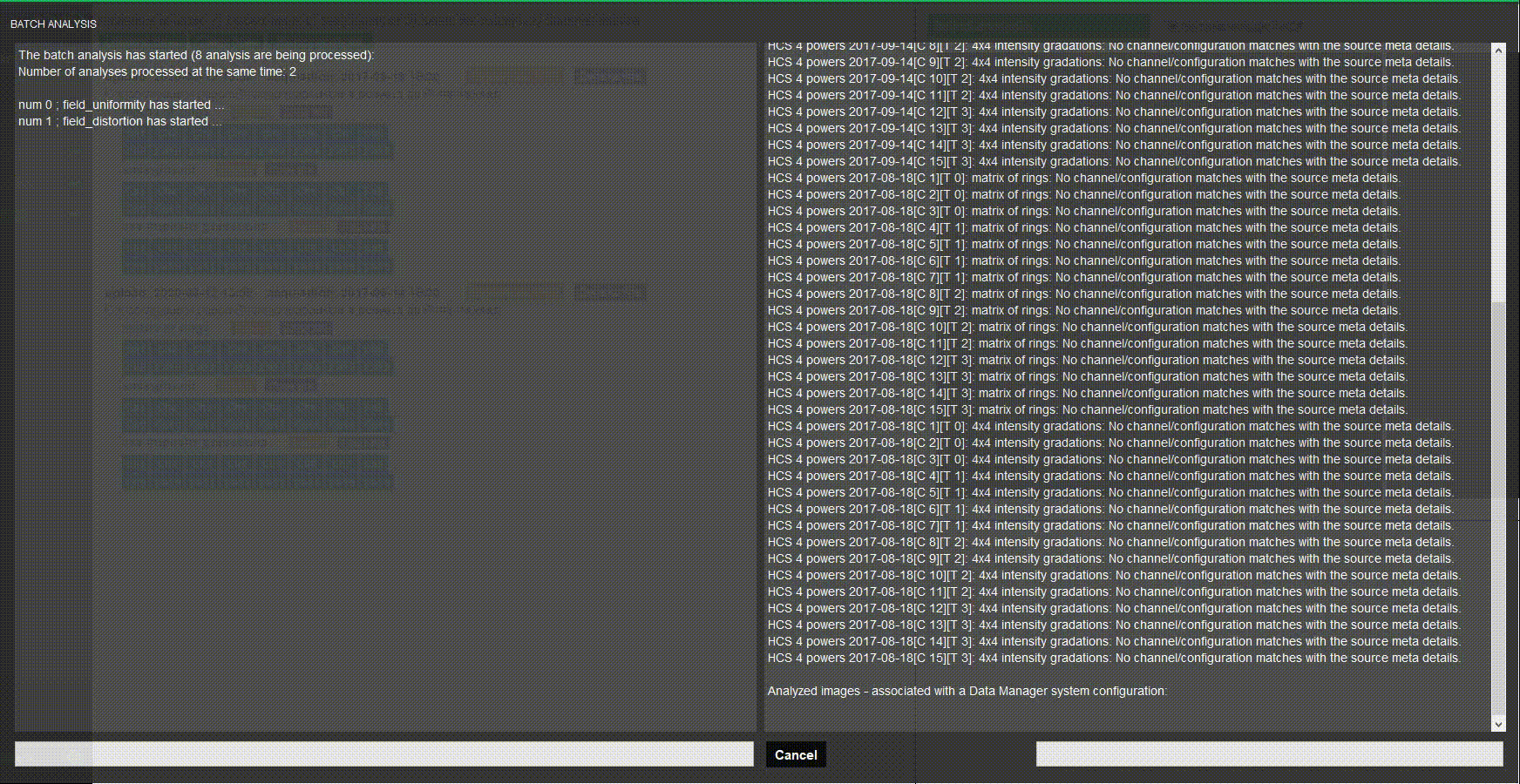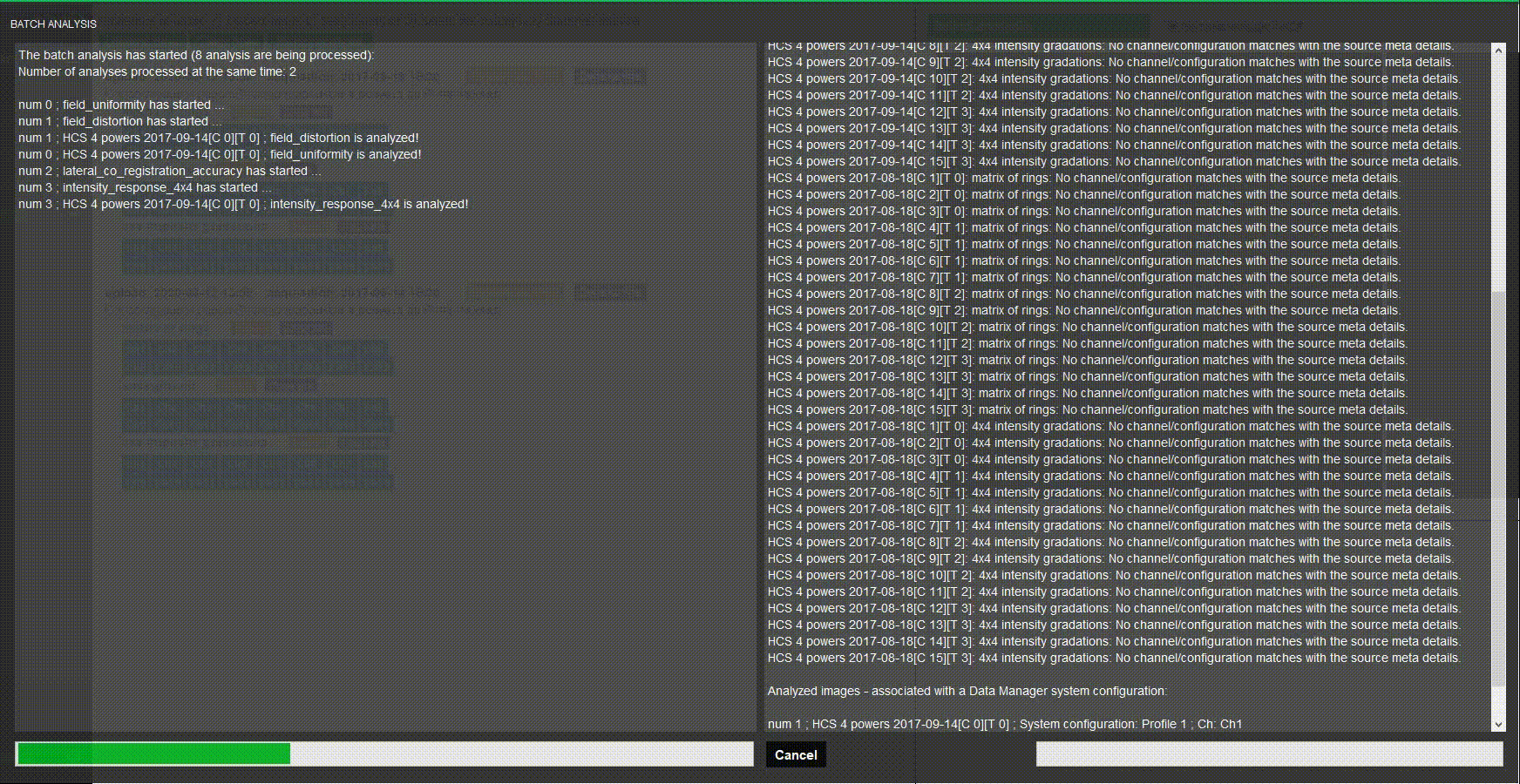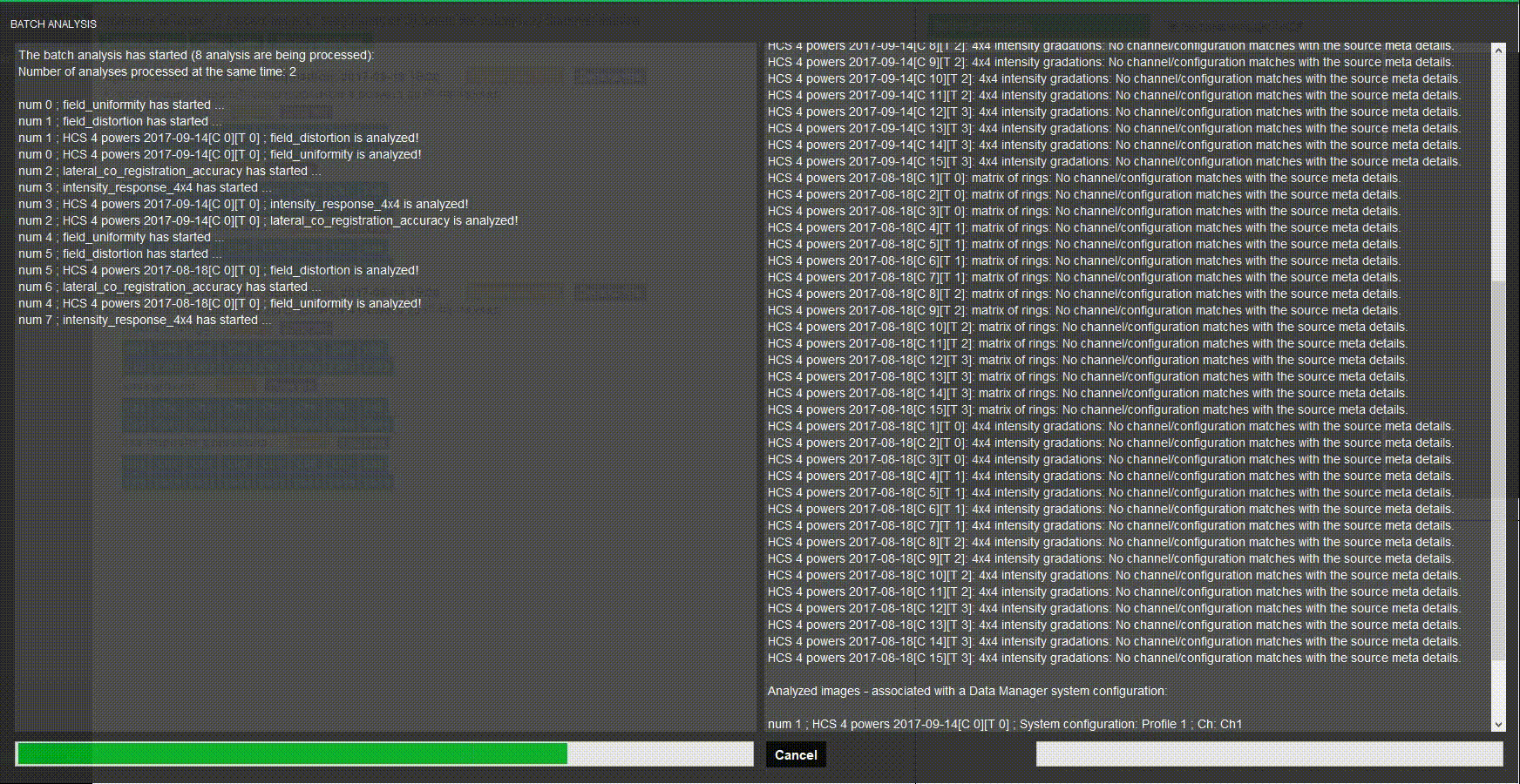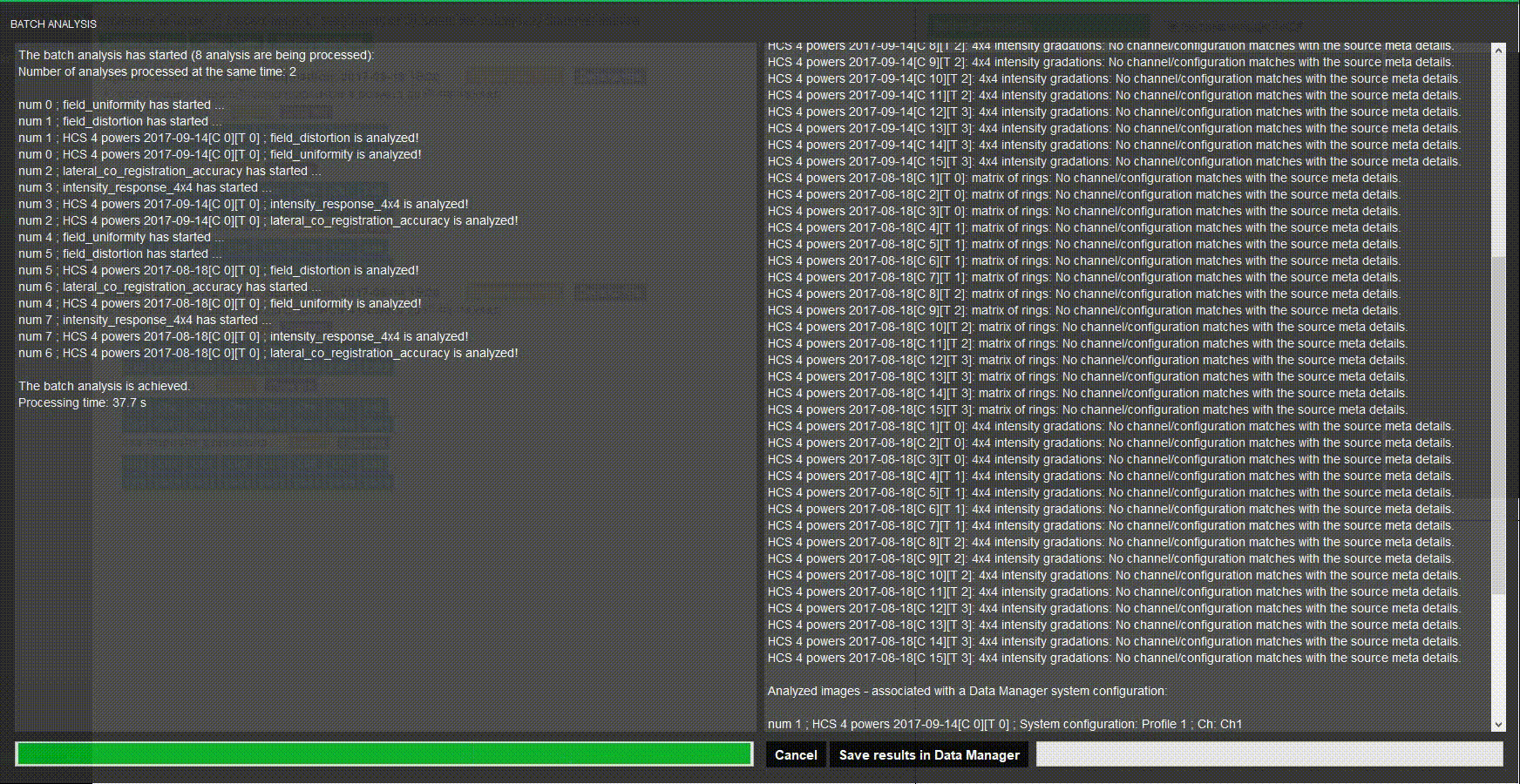
Batch process images
Do you have several systems to assess? Stop wasting time when processing images one by one. Batch processing in Daybook analyzes multiple images at once.
Choose a naming convention, load your images and there you go!

ISO Compliance
Daybook software is compliant with the ISO 21073:2019 norm entitled “Optical data of fluorescence confocal microscopes for biological imaging” > Learn more about ISO 21073
Licensing
With one license, multiple users have the flexibility to access their database accross multiple computers. One microscope monitoring slot is provided, add more slots to monitor a fleet of systems progressively.
Choose from licensing options spanning one or five years. Inclusive maintenance and updates run throughout the entire license period, guaranteeing that your software remains cutting-edge and optimized.
Automated analyses
Run more than 12 automated analyses to measure field, chromatic aberrations.
Using images
Load images for multiple format (*.CZI, *.LIF, *.ND2, *.IOR, etc)
Monitoring
Monitor several systems with different configuration and track it over time.
Thresholds
Create your own tolerance thresholds to detect issues. Add comment to get static information.
Batch analysis
Analyze a batch of images at once using keyword to match analyses.
Database
Get relevant and comparable values and save then in an organized database.
Reports
Explore, share and report on the performance of your different system.
Support & Update
Online support and frequent updates.
MINIMAL CONFIGURATION:
Screen resolution: 1280×720
64 bits operating system and processor required
Operating system: 64-bit Windows 8.1, Windows 10, Windows 11
RAM: 4 GB
Network: High-speed internet connection
Free hard drive space: 10 GB
RECOMMENDED CONFIGURATION:
Screen resolution: 1920×1080
64 bits operating system and processor required
Operating system: 64-bit Windows 8.1, Windows 10, Windows 11
RAM: 8 GB
Network: High-speed internet connection
Free hard drive space: 20 GB
The application executes https requests to https://api.daybook.fr and https://account.daybook.fr. It is mandatory to authorize these requests through the firewall if needed.
This application is not designed to work on a virtual machine.